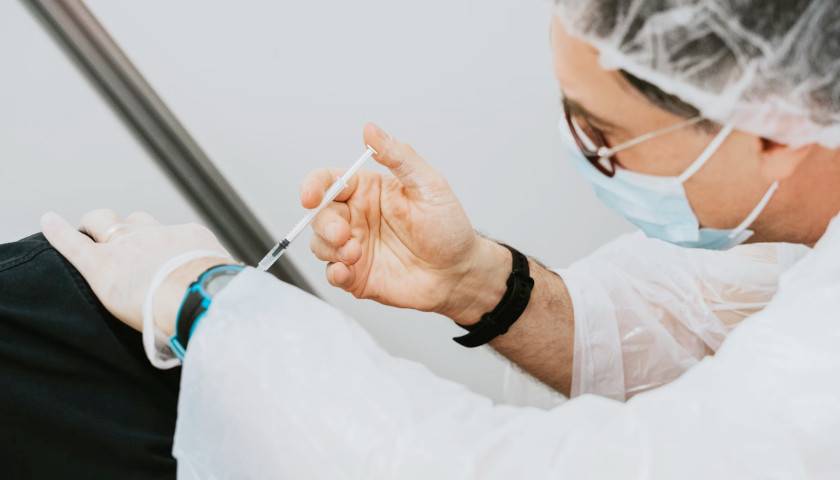
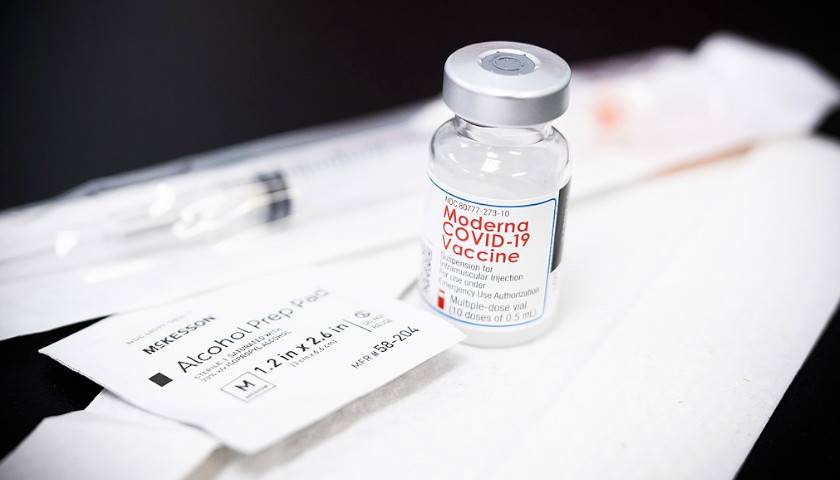
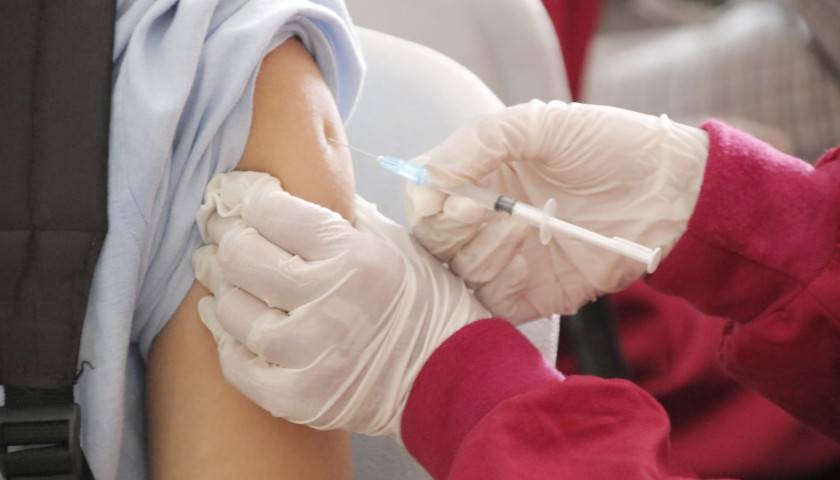
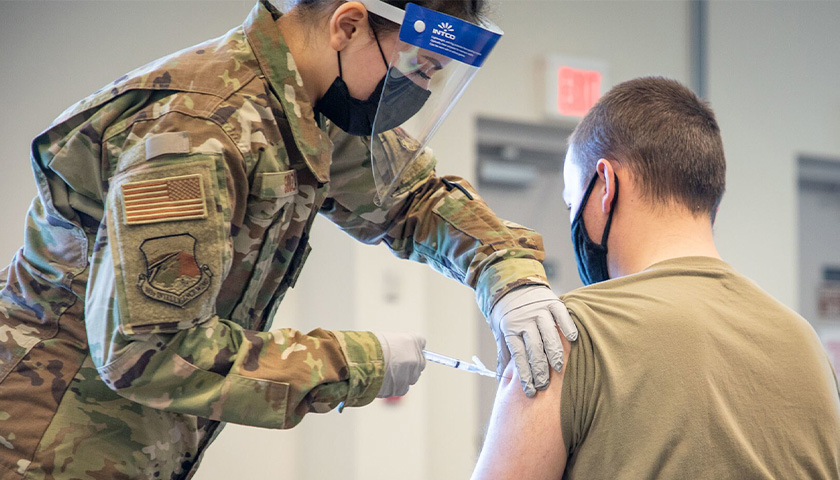
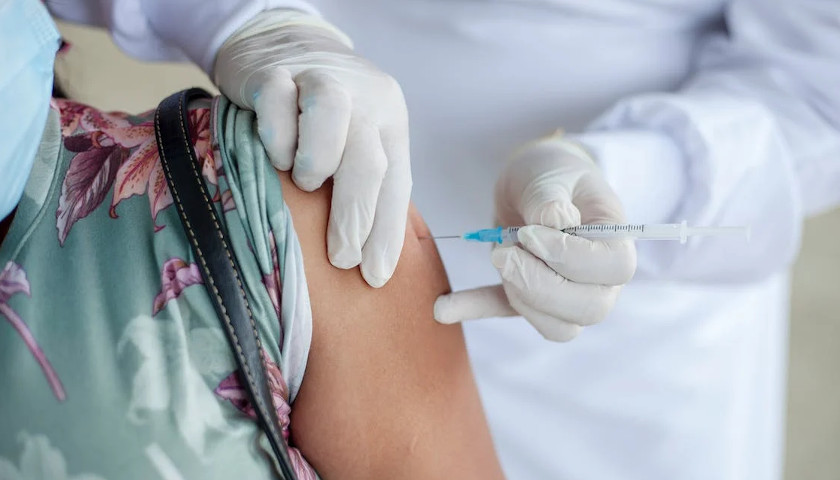
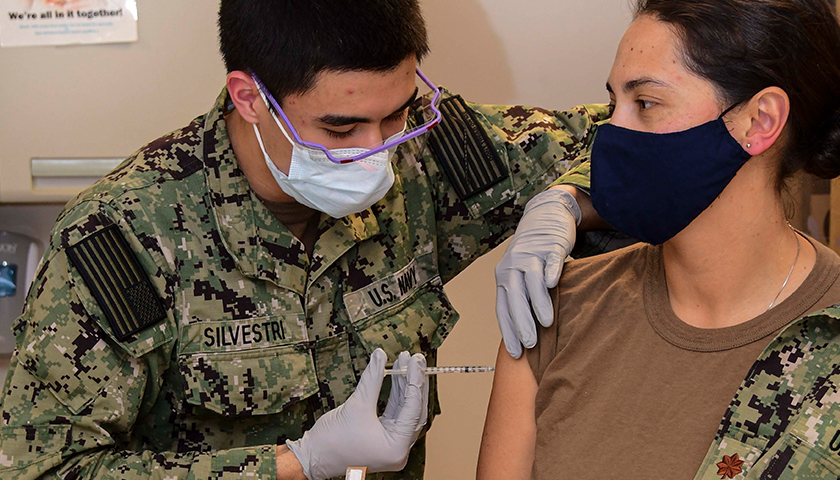
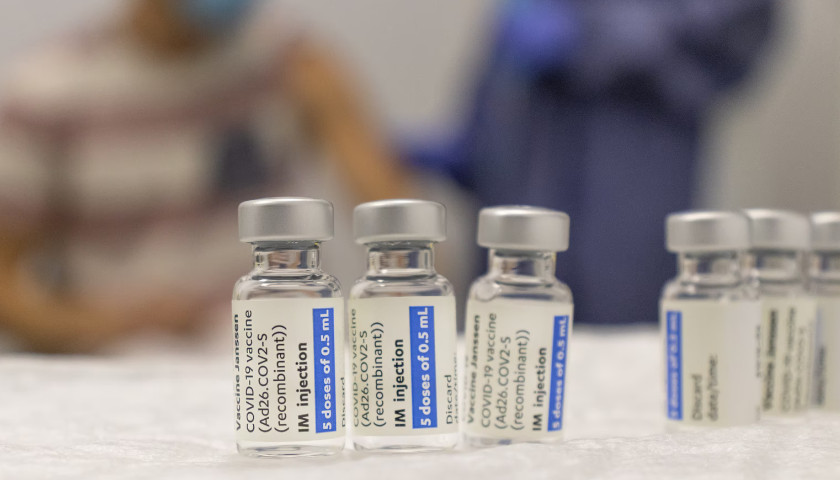
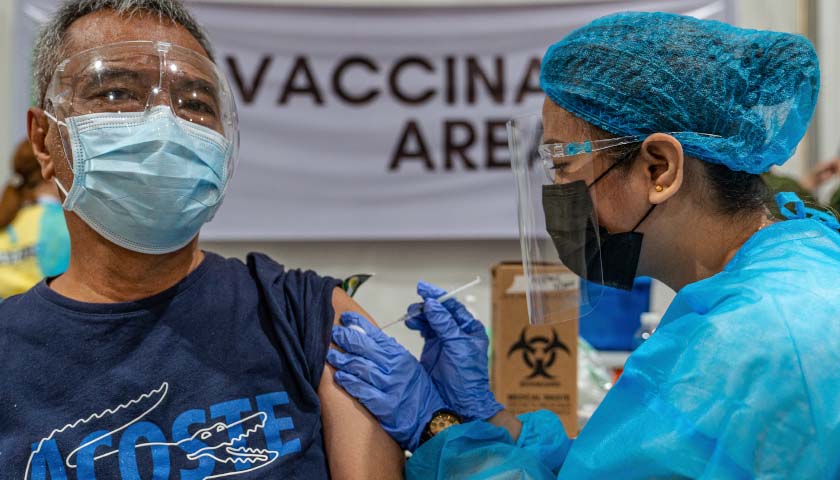
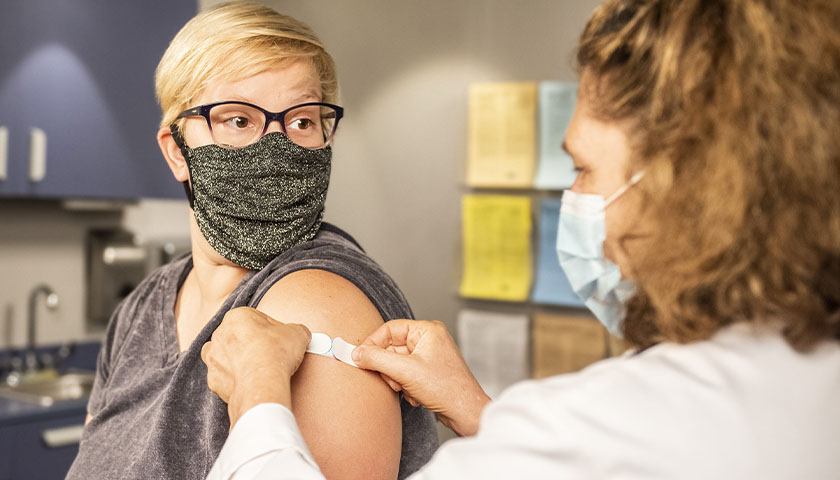
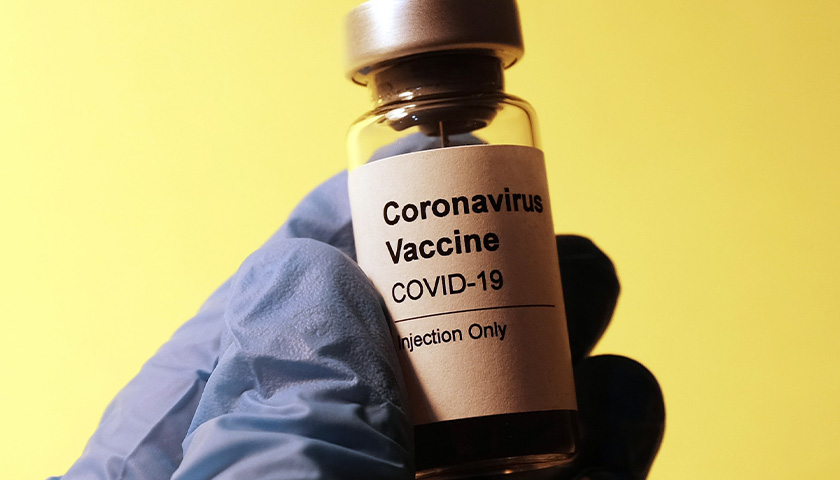
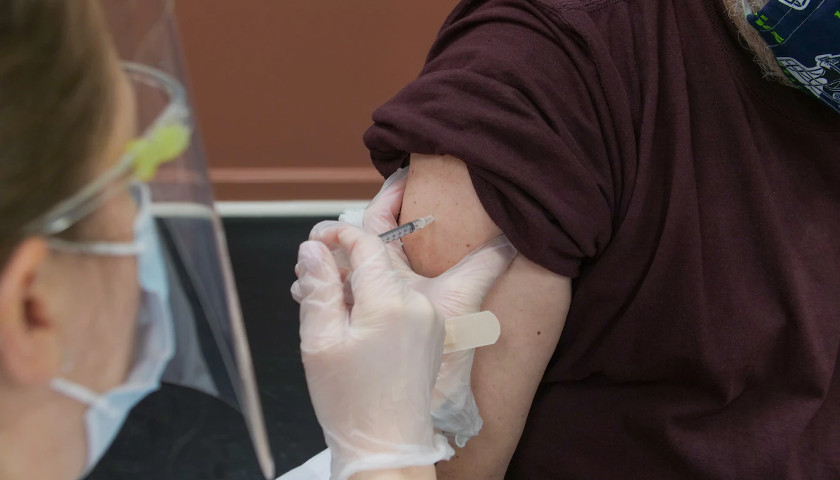
The earliest recipients of newly authorized COVID-19 vaccines, including healthcare workers, wrote in tens of thousands of adverse events related to the heart, ears, reproductive system and other conditions not listed as checkboxes in a federal active monitoring smartphone app.
The Centers for Disease Control and Prevention in the past two months turned over 780,000 “free text” entries from V-safe, the agency’s vaccine-safety monitoring system, under a January order by U.S. District Judge Matthew Kacsmaryk in a Freedom of Information Act lawsuit by Freedom Coalition of Doctors for Choice.
Read More